
Product compliance management software helps food and manufacturing companies verify supplier documentation, control product release decisions, and maintain audit ready traceability. In highly regulated industries, even small documentation gaps such as an unchecked Certificate of Analysis (COA) or an unresolved supplier deviation can lead to recalls, audit failures, and regulatory penalties.
Modern product compliance management systems centralize COA verification, supplier approvals, non conformance tracking, and corrective action workflows within one structured platform. Instead of relying on spreadsheets, emails, or paper based records, teams gain real time visibility and a secure, timestamped audit trail for every compliance decision.
This guide explains how product compliance management software works, the key features to evaluate, and how the right system reduces regulatory risk while strengthening supplier accountability and improving operational efficiency.
Do You Actually Need This? A Quick Diagnostic
Not every operation needs a dedicated compliance platform. Here are six signals that yours has outgrown spreadsheets and email:
- Manual COA cross-referencing. If anyone on your team compares certificate values against specification parameters by hand, one oversight is all it takes for a non-compliant batch to move through.
- The product takes more than 24 hours to clear. Manual verification routinely turns routine holds into multi-day delays. If QA is the bottleneck on every incoming shipment, the problem is process design, not headcount.
- A non-conformance was caught late, or not at all. If a supplier deviation reached production because a COA check was missed or misread, your current system has a gap that needs closing.
- Preparing for GFSI, SQF, BRC, or FSMA certification. These standards require documented, traceable evidence of incoming material verification and release decisions. Assembling that evidence from email threads before an audit is a costly and avoidable problem.
- Supplier base growing past 15 to 20 vendors. A handful of suppliers managed manually is manageable. Twenty-five, with varying COA formats and different document requirements per material, is a different problem entirely.
- No visibility into supplier non-conformance trends. If a vendor has had repeated deviations and no one in your organization has a clear picture of the pattern, sourcing decisions are being made without the evidence to support them.
If three or more of these apply,
A dedicated compliance platform is likely worth evaluating in detail. Fewer than three may indicate that improving your existing QMS or document management system could be the more practical first step.
What Product Compliance Management Software Actually Does
Here is the complete end-to-end workflow a purpose-built compliance platform runs in food and manufacturing operations, from defining release requirements through to alerts and notifications:
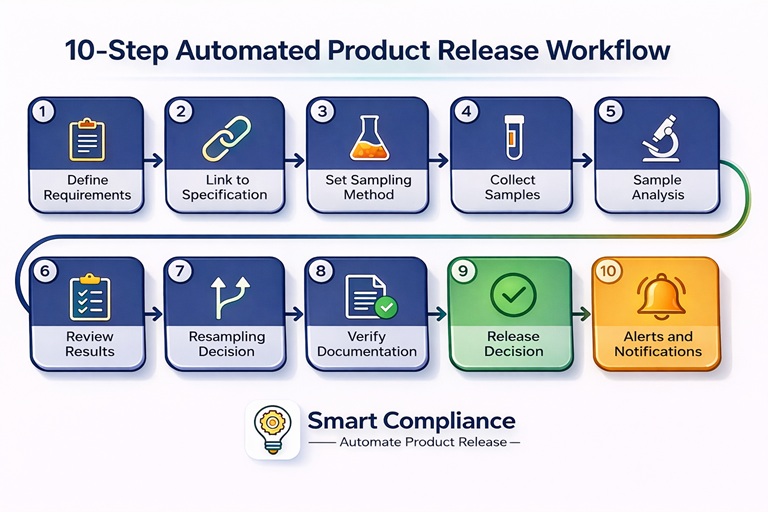
- Define product release requirements: Establish the criteria and standards for product release, BOL, COA, packing list, origin certificate, and any other regulatory or internal requirements applicable to that product.
- Link to product specification: Align detailed release criteria with the product specification, ensuring every verification check draws from the correct, approved version of the spec.
- Set sampling method and sample size: Determine the appropriate sampling method and sample size to representatively assess compliance for each incoming lot.
- Collect samples: Capture samples using the defined protocol, logged and traceable within the system.
- Perform sample analysis: Analyse collected samples against the product specification. Results are entered or imported directly into the compliance record.
- Review analysis results: Every parameter is reviewed against specification tolerance. Compliant parameters are confirmed; deviations are flagged automatically with the relevant non-conformance workflow triggered.
- Resampling decision: If a deviation is identified, the system determines whether additional sampling is required, either as an extension of the original sampling plan or as a standalone resampling process, before a release decision is made.
- Documentation verification: All documentation and records related to sampling, analysis, and compliance management are verified for completeness and accuracy.
- Release decision: An informed product release or hold decision is generated based on the full analysis results and documentation verification, with the decision logged and timestamped.
- Alerts and notifications: Relevant stakeholders receive timely alerts on compliance status, deviations, hold decisions, or any required actions, keeping the right people informed without manual follow-up.
In practical terms:
In high-volume operations, incoming material verification involves a large number of individual parameter checks across multiple shipments every day. A compliance platform runs those checks automatically, flags exceptions in real time, and maintains a complete audit trail without manual cross-referencing.
The Features That Actually Matter
Most competing articles mention user friendly interface and customization features. These are just the minimum things that one can expect, not the things that make the product different from others. The features that really distinguish a good compliance platform are,

Specification Version Control
Product specs change because of new rules, product changes, or customer needs. So, use a platform that tracks spec versions. It should link the right version to each order. This stops errors from using outdated specs to check incoming COAs. Make sure it has approval steps for spec changes and auto version updates for new deals.
COA Parsing Across Multiple Document Formats
Suppliers do not standardise their COA formats. Some send structured PDFs, some export from their lab software, some send Excel files. Flexible document ingestion that can extract values from varied supplier layouts without manual reformatting is critical for any operation with a diverse supplier base.
Configurable Tolerance Bands and Conditional Release Logic
Compliance in practice is rarely a simple pass or fail. A moisture reading 0.2 percent over limit may warrant a conditional hold; the same deviation at 2.0 percent over requires immediate rejection. A strong platform supports configurable escalation thresholds and conditional release logic rather than binary outcomes only.
Supplier Document Requirements Per Transaction
Different suppliers and materials require different documentation packages. A compliance platform should configure and track these requirements at the transaction level automatically, covering COAs, Bills of Lading, packing lists, origin certificates, food safety certifications, and any other documents required per supplier or material type.
CAPA Closure With Full Documentation
Logging a non-conformance is not the same as managing it. A strong platform assigns Corrective and Preventive Actions to vendors with defined response deadlines, tracks vendor responses, and closes CAPAs with documented evidence, building a supplier management record rather than just a deviation log.
Supplier Non-Conformance Trending
One non-conformance is an incident. Ten from the same supplier over twelve months is a risk profile. The platform should aggregate deviation data by supplier and parameter type so your team can have evidence-based conversations with vendors and make informed sourcing decisions rather than reacting to individual events in isolation.
Best Product Compliance Management Software in Reviewed
Each platform below is evaluated on how well it serves its stated audience, COA-to-release workflow, supplier document management, non-conformance handling, and audit readiness.
1. Smart Compliance

Smart Compliance is a product compliance platform designed around the COA verification and automated product release workflow. It was developed for food, beverage, and manufacturing operations where incoming material verification is a daily, high-volume activity, not an occasional administrative task.
When a supplier submits a Certificate of Analysis, the system compares every parameter against the current version of the product specification automatically, calculates compliance in real time, and generates a release decision without manual intervention. Non-compliant lots are held, logged, and assigned for corrective action. If additional sampling is needed, the system manages the re-sampling decision before a final release call is made.
Our system manages spec versions automatically. Each version goes through an approval process and is then used to check new transactions. This way, incoming documents are always checked against the correct, current spec. The platform also includes Smart Docs, a document control module covering approval workflows, version control, and audit readiness for SOPs, policies, and quality documentation.
Smart Compliance serves food manufacturers, seafood processors, dairy and poultry operations, pharmaceutical manufacturers, and all businesses where COA-to-spec verification and product release is a daily workflow need, in environments where preventing quality failures and minimizing supply chain disruption are critical to operations.
Key Capabilities
|
Best for Food manufacturers, beverage processors, seafood, dairy, poultry, and pharmaceutical operations where COA verification and automated product release is the core daily quality workflow.
2. ComplianceQuest
Compliance Quest is a cloud-based quality and compliance platform. It covers QMS, EHS, Supplier Relationship Management, and Product Lifecycle Management in a unified environment, designed for large, complex organisations where quality, safety, and compliance functions need to be tightly integrated with commercial and supply chain operations. It is well-established across pharmaceutical, medical device, aerospace, and automotive industries.
Key Capabilities
|
Best for Large enterprises in pharmaceutical, medical device, or aerospace sectors requiring integrated quality, safety, and compliance management at scale.
3. MasterControl
MasterControl is a cloud based quality and compliance management platform designed for highly regulated manufacturing environments. With more than two decades of industry presence, it is widely used in pharmaceutical, biotechnology, and medical device industries. The platform supports structured quality processes including document control, electronic batch records, CAPA management, audit management, and employee training tracking.
The system is built to help organizations maintain controlled workflows, secure documentation, and consistent regulatory compliance across complex production operations.
Key Capabilities
|
Best for Pharmaceutical, biotechnology, and medical device manufacturers operating under FDA or EU GMP regulatory requirements.
4. Sphera
Sphera provides enterprise sustainability management, operational risk, and product stewardship solutions for chemical, oil and gas, industrial manufacturing, and automotive sectors. Its SpheraCloud platform integrates EHS, ESG data management, and supply chain transparency tools, supported by an extensive verified regulatory and chemical reference dataset.
Key Capabilities
|
Best for Chemical manufacturers and industrial companies with significant ESG, EHS, and product stewardship compliance obligations across global supply chains.
5. iPoint Systems
iPoint Systems offers software and consulting services that help businesses meet environmental and social compliance regulations. The platform is designed primarily for electronics, automotive, consumer goods, and other manufacturing industries that need to track materials across complex supply chains.
iPoint specialises in managing substance compliance across supply chain tiers, covering REACH, RoHS, SCIP, and conflict minerals regulations.
Key Capabilities
|
Suits for Electronics manufacturers and consumer goods companies managing REACH, RoHS, and conflict mineral compliance across complex global supply chains.
6. SafetyCulture (iAuditor)
SafetyCulture is a workplace operations platform. It is built around mobile-first inspection management, enabling teams to conduct audits, capture issues, assign actions, and monitor operational data from any device in real time. It is widely used in food manufacturing for GMP inspections, facility hygiene audits, equipment checks, and safety compliance monitoring.
Key Capabilities
|
Used by operations teams needing mobile-first inspection management and real-time audit data capture on the production floor and many more.
How to Choose Product Compliance Management Software A Practical Framework
Step 1: Identify Your Primary Compliance Driver
- GFSI, SQF, BRC, FSMA, or HACCP: You need COA verification, automated product release, and audit-ready documentation built into daily operations.
- FDA 21 CFR Part 11 or EU GMP: You need electronically validated systems, electronic signatures, and regulatory submission support.
- REACH, RoHS, or conflict minerals: You need substance declaration management across supply chain tiers.
- ESG and environmental compliance: Dedicated sustainability platforms address this set of obligations.
Step 2: Ask These Six Questions in Every Demo
These questions reveal whether the platform handles your actual workflows, not just polished demo scenarios:
- Can you run a COA verification against a live product specification using my actual document format instead of a preloaded sample?
- If a COA contains three compliant parameters and one parameter that is 0.5 percent outside the defined tolerance, what does the system do step by step?
- If a product specification changes after a purchase order has already been placed, how does the system apply the correct specification version to the incoming COA?
- What would an SQF or GFSI auditor see when requesting documented evidence of product release decisions for the last 90 days?
- My suppliers send Certificates of Analysis in different formats, including scanned PDFs, Excel files, and email text. How does the system process each format without requiring manual reformatting?
- How can I view a supplier’s non conformance history over the last 12 months, and how can I export it as a structured performance report?
Step 3: Account for Full Cost of Ownership
- Implementation timeline: Platforms vary significantly in how long implementation takes, from weeks for focused tools to many months for enterprise platforms with broad scope. Factor implementation time into your total cost evaluation, not just the subscription price.
- Configuration ownership: Platforms that require IT involvement for every workflow change carry higher ongoing operational costs than those QA managers can update directly.
- Integration requirements: If the platform does not connect to your ERP, QMS or LIMS natively, custom integration is an additional project with its own timeline and budget.
- Training investment: Consider how much structured training is required before your QA team can use the platform productively. The steeper the learning curve, the longer before the platform delivers value.
Conclusion
Choosing the right compliance platform comes down to one question: what is the specific problem you need to solve every day?
For food, beverage, and manufacturing operations, that problem is incoming material verification, checking that every supplier COA matches the current product specification, generating a release decision automatically, and maintaining a complete audit trail without manual intervention. Smart Compliance is built around that workflow, from the moment a COA is received to the final release decision and supplier notification.
If you want to see the 10-step automated product release workflow in action against your actual specifications and supplier documents, book a free demo.




